Sensitivity enhancement by DNP
One of the most significant limitation of solid-state NMR is its intrinsically poor sensitivity. As results, it conventionally required a large quantity of protein sample (>1mg) for a full structural study, and was difficult to analyze huge molecular entities since the unit volume sensitivity drops with the molecular weight of individual protein.
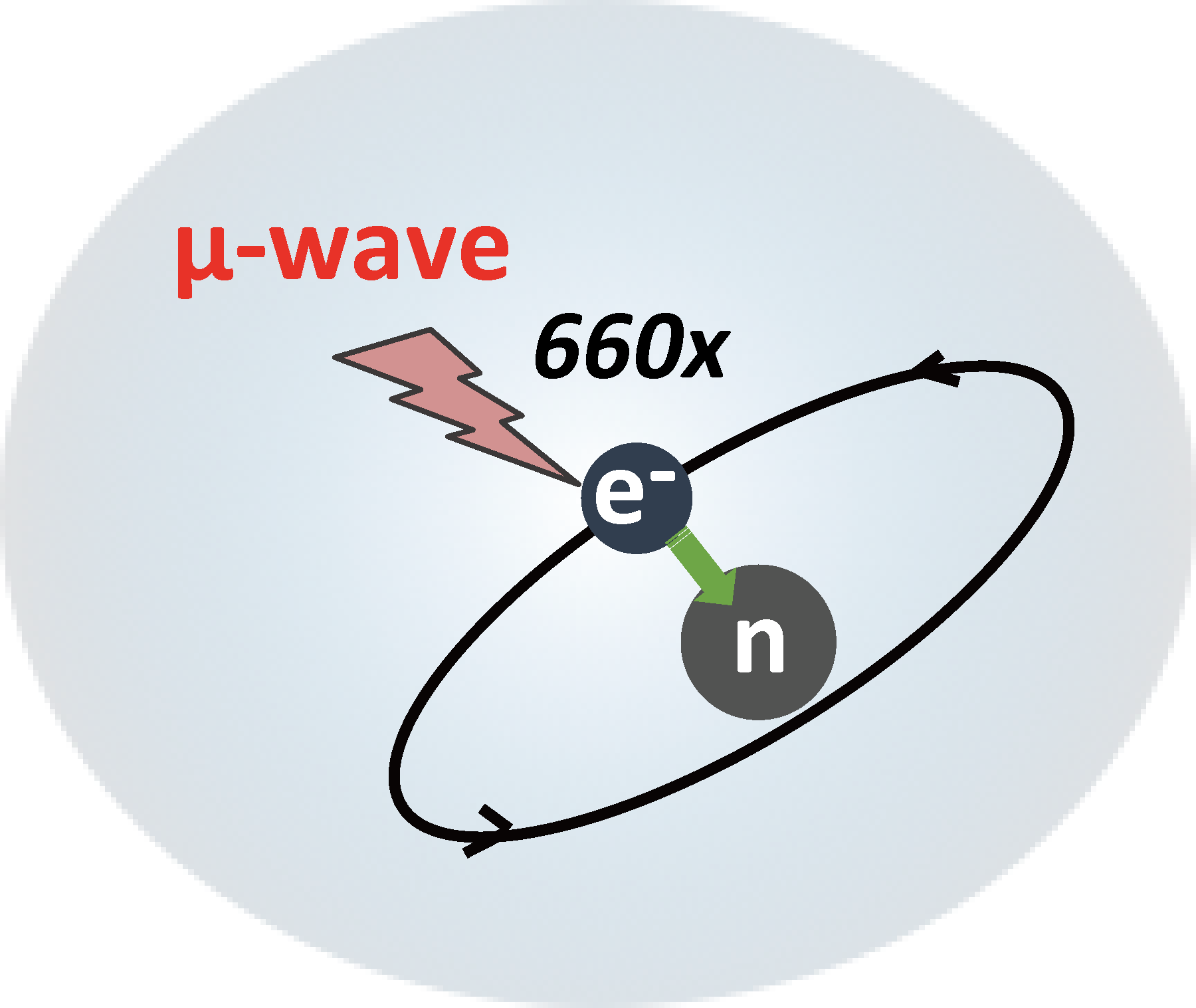
Here, we bring up the DNP. Dynamic nuclear polarization (DNP) is a method that dramatically improve the sensitivity of SSNMR by transferring the 660-times larger Boltzmann polarization of an electron to the nuclei of interest by an irradiation of high-power microwave near the EPR frequency of some paramagnetic polarizing agent co-doped with the sample of interest.
In addition to DNP, sample cooling can inverse-proportionally increase the nuclear polarization according to the Curie’s law, i.e. at 30 K (vs 300 K, room temperature), the nuclear polarization grows 10-times stronger. In combination, the total signal enhancement would become phenomenal, i.e., 660x10=6600!
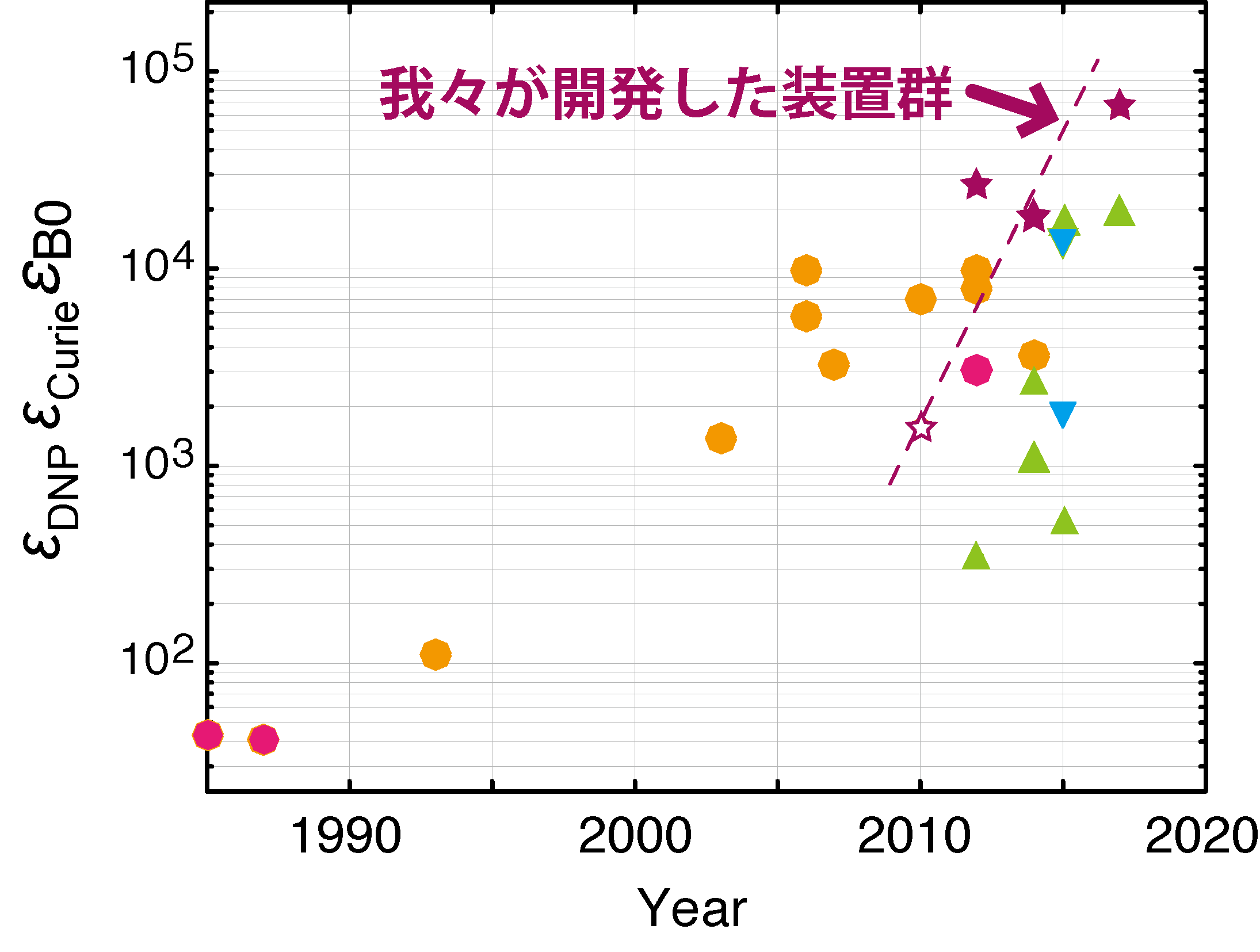
Since 2005, we have been developing instruments for high-field DNP MAS NMR spectroscopy. During this course, we have reported the world’s first over-10T DNP observation at 14.1 T in 2010, 16.4 T at 2014. Also in 2015, we commissioned the first complete closed-cycle helium MAS system for ultra-low temperature DNP at T ~ 30 K, as well as a dual gyrotron setup that enables highly flexible DNP experiments ever possible. This has yielded unprecedentedly high (~1000 folds) signal receptivity at high field conditions suitable to study big and complex chemical systems like proteins.

Our current challenge is to establish general sample prep protocols to optimize the spectral resolution at cryogenic sample temperature, and ways to stably polarize the broad range of samples such as membrane proteins, amyloid fibrils and proteins in cell, looking toward wide applications.