![]()
ACHIEVEMENTSIPR The University of Osaka
-

- ACHIEVEMENTS
- Mycoplasma mobile moves into overdrive: twin motor modified from ATP synthase discovered
Press Release
2021.07.21
Mycoplasma mobile moves into overdrive: twin motor modified from ATP synthase discovered
Title
Mycoplasma mobile moves into overdrive: twin motor modified from ATP synthase discovered
Subtitle
Scientists discover gliding machinery for Mycoplasma mobile to be a new structure that could share ancestry with ATP synthase
Summary
Using electron microscopy and high-speed atomic force microscopy, researchers show the internal molecular motor behind the gliding mechanism for Mycoplasma mobile to consist of two ATP synthase-like molecules. Sharing a similar structure with ATP synthase, suggesting a common evolutionary ancestor. This synthase-like ATPase is challenging the origin of cells and proteins themselves.
Body
For more than 20 years, Makoto Miyata from Osaka City University has been studying the gliding motility of the parasitic bacterium Mycoplasma mobile (M. mobile). A mechanism consisting of an external “grabbing” structure and internal “motor”, the motor having yet to be clarified on a molecular level. In collaboration with Osaka University and Kanazawa University, his research team used electron microscopy and high-speed atomic force microscopy (high-speed AFM) to reveal that the bacteria’s molecular motor consists of two ATP synthase-like complexes, suggesting an unexpected evolution of the protein. Their findings were published in mBio.
Based on genetic information, researchers have suggested that the molecular motor of M. mobile evolved from a protein that synthesizes ATP, called ATP synthase, although this had yet to be confirmed. ATP is an important substance generated in the mitochondria that provides energy that a molecular motor uses for the movement of nearly all living things, including M. mobile gliding.
“It was recently hypothesized that M. mobile, unlike respiration systems in mitochondria, converts ATP energy into a gliding force with a rotary class of ATP synthase,” said Makoto Miyata. “To our surprise, we discovered the motor of M. mobile to be a chain of two ATP synthases-like molecules, housed within a complex cylindrical structure,” he continued – something that has never been seen before.
The discovery started with the team extracting a cell from M. mobile and isolating its protein units. Using negative-staining electron microscopy, they discovered a twin motor where each motor is structurally similar to ATP synthase. However, the clarification has not stopped here. “We need to see this dimeric ATP synthase chain in action,” stated Noriyuki Kodera of Kanazawa University, “to understand how it facilitates M. mobile’s gliding movement.” Using the state-of-the-art technology high-speed AFM, pioneered at the Nano Life Science Institute of Kanazawa University, the team plans to confirm the rotary nature of the molecular motor during ATP hydrolysis. As the architecture of the gliding motor is like ATP synthase, “it is possible the chains share a common evolutionary origin,” stated Takuma Toyonaga, a research fellow at Osaka City University.
Toyonaga continued, “This discovery of an ATP synthase created a breakthrough in our understanding of the evolution of cells and protein and has brought us closer to understanding the operating principles of motility, which is expected to serve as a basis for the development of nanoscale devices and pharmaceuticals.”
###
Funding
- Grants-in-Aid for Scientific Research on Innovative Areas “Harmonized supramolecular machinery for motility and its diversity
- Grants-in-Aid for Scientific Research (A) “Three types of motility mechanisms in pathogenic bacteria, class Mollicutes”
- Grants-in-Aid for Scientific Research (B) “Structure and movement of gliding machinery for Mycoplasma gliding”
- CREST: “Construction of a cell evolution model using synthetic bacterium JCVI syn3.0B and genome manipulation” (Principal Investigator: Miyata)
https://www.jst.go.jp/kisoken/crest/en/project/1111100/1111100_2019.html - Strategic Research Funds for Osaka City University, FY2018 (Priority Research)
- Fugaku Fund for Biomedical Research (Research Representative: Makoto Miyata)
- Grants-in-Aid for Scientific Research and Specially Promoted Research
“Elucidation of Three-Dimensional Structure and Function of Biomolecular Motors by Cryo-Electron Microscopy - Ministry of Education, Culture, Sports, Science and Technology Life Science Research Support Platform for Drug Discovery, etc.
(Platform project for supporting technology platform for drug discovery, etc.) - Japan Agency for Medical Research and Development “Medical Research and Development Innovative Platform Project”
- Japan Medical Research Organization “Medical Research and Development Innovative Technology Platform Project” – JEOL YOKOGUSHI Collaborative Research Institute (Research representative: Keiichi Namba, Professor, Osaka University)
Graphic
Title:
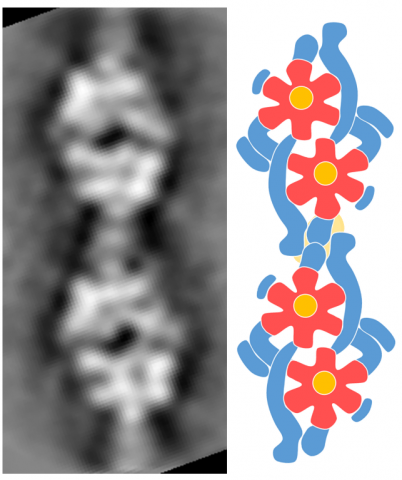
Gliding motor
Caption:
Two sets of twin motors are connected. An image reconstructed from electron microscopy (left) and a schematic (right) are shown. The flower like structure colored red and yellow shares its structure with ATP synthase, which is widely spread among organisms.
Credit:
Toyonaga T, Kato T, Kawamoto A, Kodera N, Hamaguchi T, Tahara YO, Ando T, Namba K, Miyata M.